Research Governance
All research activities, no matter how big or small, have implications in terms of ethics, safety, time and resources, and for this reason require oversight (otherwise known as governance) to ensure the relevant legislative, regulatory and policy requirements are met and activities align with the values of the organisation. In Northern NSW Local Health District, we encourage research to be undertaken in areas that are of strategic merit and which could lead to meaningful change. Please refer to the Northern NSW Local Health District Research Principles for a list of these strategic areas.
On this page:
What is Research Governance?
Research Governance is a framework through which institutions are accountable for the scientific quality, ethical acceptability and safety of the research they sponsor or permit. Research Governance, including both ethics and site authorisation processes, ensures compliance with a broad range of regulations, legislation and codes of good practice to achieve and continuously improve research quality across all aspects of healthcare by:
- Safeguarding the dignity, rights, safety and well-being of participants
- Protecting and promoting the integrity of research and investigators
- Enhancing ethical and scientific quality
- Minimising risk
- Monitoring practice and performance and
- Promoting good practice
- The Research approval and authorisation process
Research Governance consists of two key processes:
Ethics and scientific review
Site Specific Authorisation or an Access Request
1. Ethical and scientific review
All research conducted within Northern NSW (NNSW) Local Health District (LHD) and/or the Mid North Coast (MNC) LHD involving participants (e.g. patients including their health information, or health service employees) must undergo ethical and scientific review and obtain approval before commencing. This review ensures research is conducted in accordance with the National Health and Medical Research Council (NHMRC) National Statement on Ethical Conduct in Human Research (2007) (the National Statement).
At Northern NSW LHD, varying levels of ethics review have been implemented. These reviews depend upon the type and level of risk associated with the activity, otherwise known as proportional risk review pathways. Risk is assessed in terms of the type of harm (physical, psychological, social, economic and legal), severity of the harm and potential for it to occur. Research activities are categorised as either:
- Negligible risk: where there is no foreseeable risk of harm or discomfort; and any foreseeable risk is no more than an inconvenience to participants. Examples of inconvenience in human research include filling in a form, participating in a de-identified survey or giving up time to participate in a research activity.
- Low risk: where the only foreseeable risk is one of discomfort. Discomforts include, for example, minor side-effects of medication, discomforts related to measuring blood pressure and anxiety induced by an interview.
- Greater than low risk: where the risk, even if unlikely, is more serious than discomfort, the research is not low risk.
Research projects of any risk can be reviewed by an appropriately constituted human research ethics committee (HREC). Low or negligible risk projects can also be reviewed by an Executive Committee of the HREC or by a non-HREC level in accordance with the National Statement. Greater than low risk projects can only be reviewed by a fully constituted HREC.
The non-HREC pathway, as noted above, is for the review of research activities of negligible or low risk. This review pathway is available to NNSWLHD employees and includes review and approval to undertake:
- Quality Activities, including retrospective analyses
- Case studies
These activities are reviewed and approved by the Director of Research or their delegate, to be consistent with the National Statement on Ethical Conduct in Human Research (2007).
The HREC pathway is for the review of research activities of negligible, low or greater than low risk. These activities must be submitted through the Research Ethics Governance Information System (REGIS). Review and approval of these research activities is undertaken by a HREC in accordance with the National Statement.
Depending on the size and type of your research activity, ethics approval can be obtained from the following HRECs:
- single-site within NNSW or MNC LHDs from the North Coast NSW HREC,
- multiple-sites within NSW from a ‘Certified NSW HREC’, or
- multiple-sites nationwide from a ‘Certified HREC’ under the National Mutual Acceptance
2. Site Authorisation
Research projects involving the actual conduct of research at Public Health Organisation (PHO) sites must submit a Site Specific Assessment (SSA) application to the PHO for review and this application must be approved prior to commencing. Applications must be submitted using the purpose-built SSA form in REGIS for all new projects. The REGIS website provides guidance on how to complete the SSA application form and details the supporting documents required for making a submission.
A separate SSA application must be made for each site at which research is to be conducted. For example, even if the project is to be conducted at two sites under the control of a single NSW PHO, a separate application must be made for each site.
Access Request (AR)
An AR application is most appropriate when the Coordinating Principal Investigator responsible for the research project is requesting support from a NSW PHO (e.g. NNSWLHD) in the form of access to participants, tissue or data but does not require the involvement of the PHO to conduct the research.
Application Form for Access Request
The NNSWLHD will generally accept an AR application (over a SSA application) on the understanding that the project meets the following criteria:
- Approval from an NHMRC Registered HREC has been obtained;
- The LHD will not be conducting the research (i.e. prospective data collection or analysis);
- Data supplied by the LHD will be non-identifiable unless participant consent is obtained by the research team1;
Examples of research activities conducive to the AR pathway are as follows:
- promoting research opportunities to potential participants by advertisement (e.g. flyer, poster) or email/letter of invitation;
- distribution of invitation to LHD employees/agents to participate in a research activity (e.g. survey, focus group, interview);
- access to data or tissue held by the LHD but not processing or analysis.
1 In accordance with the Information and Privacy Commission New South Wales Statutory Guidelines on Research – Health Records and Information Privacy Act 2002 (NSW) dated 01.09.2004
Quality Activities
There are several approaches for monitoring and evaluating healthcare with the aim of improving health service delivery. Some of these approaches can be broadly categorised as Quality Activities (QAs), and include Quality Improvement (QI), Quality Assurance, and program/service evaluation, see figure 1.
Figure 1: Continuum of research activities
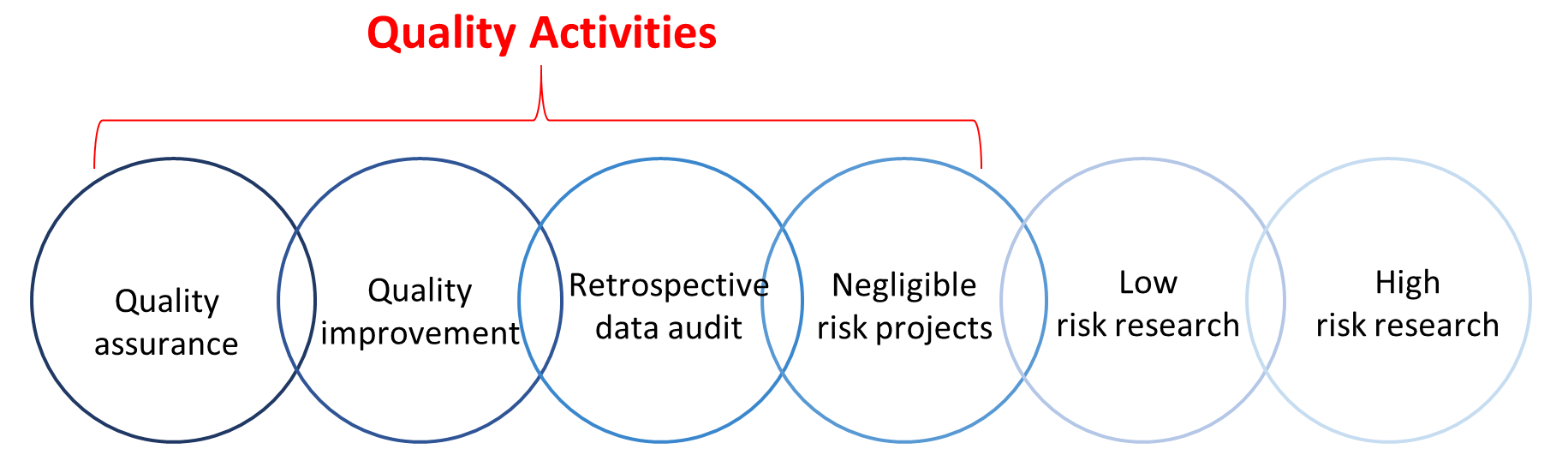
While attempts to differentiate QAs as separate to research by way of definition are generally unhelpful (because they often utilise similar methods), the following terms broadly describe how research and different types of QAs may be viewed:
-
- Research: The creation of new knowledge and/or the use of existing knowledge in a new and creative way so as to generate new concepts, methodologies, inventions and understandings. This could include synthesis and analysis of previous research to the extent that it is new and creative. 1
- Quality Improvement: Improve the performance of a health service, or program, as judged by accepted standards in accordance the current evidence.
- Quality Assurance: de-identified data analysis for the purpose of maintaining standards or identifying areas for improvement in the environment from which the data were obtained. This includes: incident monitoring, root cause analysis, sentinel event monitoring, peer review, morbidity and mortality review and other forms of audit.
- Service/Program Evaluation: Evaluation is a term that generally encompasses the systematic collection and analysis of information to make judgements, usually about the effectiveness, efficiency and/or appropriateness of an activity. The term is used in a broad sense to refer to any set of procedures, activities, resources, policies and/or strategies designed to achieve some common goals or objectives. 2
1 Australian Research Council. Accessed 20 July 2020: https://www.arc.gov.au/sites/default/files/minisite/static/10419/ERA2015/intro-3_define-research.html
2 Australasian Evaluation Society Incorporated Guidelines for the Ethical Conduct of Evaluations, August 2010: https://www.aes.asn.au/images/AES_Guidelines_web_v2.pdf?type=file
As there can be difficulty in, and disagreement about, clearly defining ‘research’ versus QA, the NNSWLHD Research Office has developed guidelines to assist staff undertaking activities that fall within the QA space to obtain appropriate oversight, including identifying when ethical considerations may be present. Specific guidelines have been developed for QAs involving:
- Quality Activity Proposal for projects involving routinely collected data (Word, 67KB)
- Quality Activity Proposal for projects involving primary data collection (Word, 70KB)
- Quality Activity Proposal for projects involving mixed methods (Word, 73KB)
The process is underpinned by a non-Human Research Ethics Committee (non-HREC) level of review and is facilitated by several resources designed to assist staff, including relevant QA proposal templates, and an ethical considerations checklist (Appendix A in all QA proposal templates), which describes common ethical considerations. The review process provides oversight in a manner that ensures activities are well-designed, and scientifically and ethically sound.
The pathway also provides a process for staff to have the Research Office formally confirm their projects are QAs to satisfy the requirements of journal editors in case they would like to submit their studies for publication in peer reviewed journals.
To request a non-HREC level of review for a QA, please download the relevant QA guideline (retrospective data analyses, primary data collection or mixed methods) on the Research Resources and Forms page and complete the following:
- Complete the relevant QA proposal template
- Complete the Ethical Considerations checklist provided at Appendix A
- Prepare/Collate any additional study material (e.g. survey tool, Participant Information Statement, Consent Form)
- Obtain endorsement for the conduct of the activity from your relevant Head of Department
- Submit the form and supporting material by email to: NNSWLHD-Ethics@health.nsw.gov.au
Please be advised that employees of Northern NSWLHD may also require approval to submit manuscripts for publication in peer reviewed journals or abstracts for presentation at scientific conferences from the Chief Executive or delegate.
Amendments to approved QA projects
Projects don’t always go to plan and changes to the protocol are sometimes required. Before making any changes to a previously approved QA project, the Lead Project Officer is required to notify and obtain Research Office approval before implementing an amendment. Please notify the Research Office of any changes you intend to make to your project which are likely to affect to a significant degree:
- The rights, safety and welfare of the participants of the project,
- The scientific value of the project,
- The conduct or management of the project, and/or
- Ethical considerations (e.g. informed consent).
To request an amendment, please complete the following:
- Provide a brief description of modifications and rationale including any summary of changes, any implications for the ongoing conduct of the project, and any need to provide new or amended project documentation.
- Update the previously approved QA Proposal and any relevant project documentation with tracked changes and updated version control in the document footer (version number and date). Note: new study documents should be submitted as version 1.
- Submit revised documents(s) to: NNSWLHD-Ethics@health.nsw.gov.au. Please include the original QA approval number in the Subject Line of the email.
Case Study / Case Series
What is a case study / case series?
A case study or report can be considered the detailed presentation of the clinical description and findings of a patient based on a hospital stay, episode of care or healthcare journey for a health condition or problem. The report itself will comprise entirely of information and data gathered during routine (standard) care delivered to a patient. A case series presents the same type of information as a case study, but for a small group of patients.
Instructions for completing and submitting a case study application for review by the Research Office
Please click-on the following link to open and save the case study/series application form. The form provides brief instructions on the requirements for completing the application, including obtaining patient consent and Head of Department / line manager support.
Application form for case study/series
To submit an application for a case study for consideration by the Northern NSWLHD Research Office, please complete the following using the application form:
- A cover letter detailing the background and rationale for the case report/study, whether it is intended that the report be submitted for publication and/or presented, and how data for the study will be accessed and stored (template provided at Appendix 1 of the application form).
- Adapt the template at Appendix 2 to obtain explicit, informed consent from the patient. The template needs to be adapted to contain the information for the coordinating investigator and their clinical unit/department. Once consent is obtained, a copy is to be submitted to the Research Office for final authorisation.
- Adapt the template at Appendix 3 for revocation of consent, which provides a formal mechanism for a patient to withdraw from a study at any time should they wish.
- Submit all completed documentation to NNSWLHD-Ethics@health.nsw.gov.au.
Please complete documents electronically (hand-written forms will not be accepted).